Cancer is one of the most complex and instructive diseases in humanswhich represents a dramatic disruption of normal cellular processes that allow cells grow uncontrollably, resist death, invade surrounding tissue and metastasize. With The incidence of cancer is increasing worldwideunderstanding its underlying mechanisms is essential for students, researchers and clinicians. Decades of research have revealed that cancer can be understood through distinct but interrelated conceptual dimensionsincluding:
-
Acquired functional abilities (characteristics of cancer)
-
Enables phenotypic traits
-
Hallmark-carrying cells in the tumor microenvironment (TME)
-
Systemic interactions within the host
Together, these dimensions give a framework for studying cancer as a dynamic, adaptive “outlaw organ”guide the development of therapeutic strategies.
Introduction: The Hallmarks concept
The concept of “characteristics of cancer” was introduced by Douglas Hanahan and Bob Weinberg to organize the great diversity of genetic and phenotypic changes observed in tumors. The aim was to understand how cancer occurs through multistep tumorigenesisdevelop through selective pressure, and acquire characteristics such as metastases, therapy resistance and immune evasion.
Timeline for characteristic development:
-
2000: Six hallmarks: maintain proliferative signaling, avoid growth suppressors, resist cell death, replicative immortality, angiogenesis, invasion/metastasis.
-
2011: Added deregulated metabolism and immune evasion.
-
2022: Phenotypic plasticity added, emphasizes dynamic adaptation to therapy and environment.
That was a key realization mutant cancer cells alone do not define tumor biology. Instead, cancer progression depends on recruitment and reprogramming of surrounding normal cellscreates one supportive tumor microenvironment (TME).
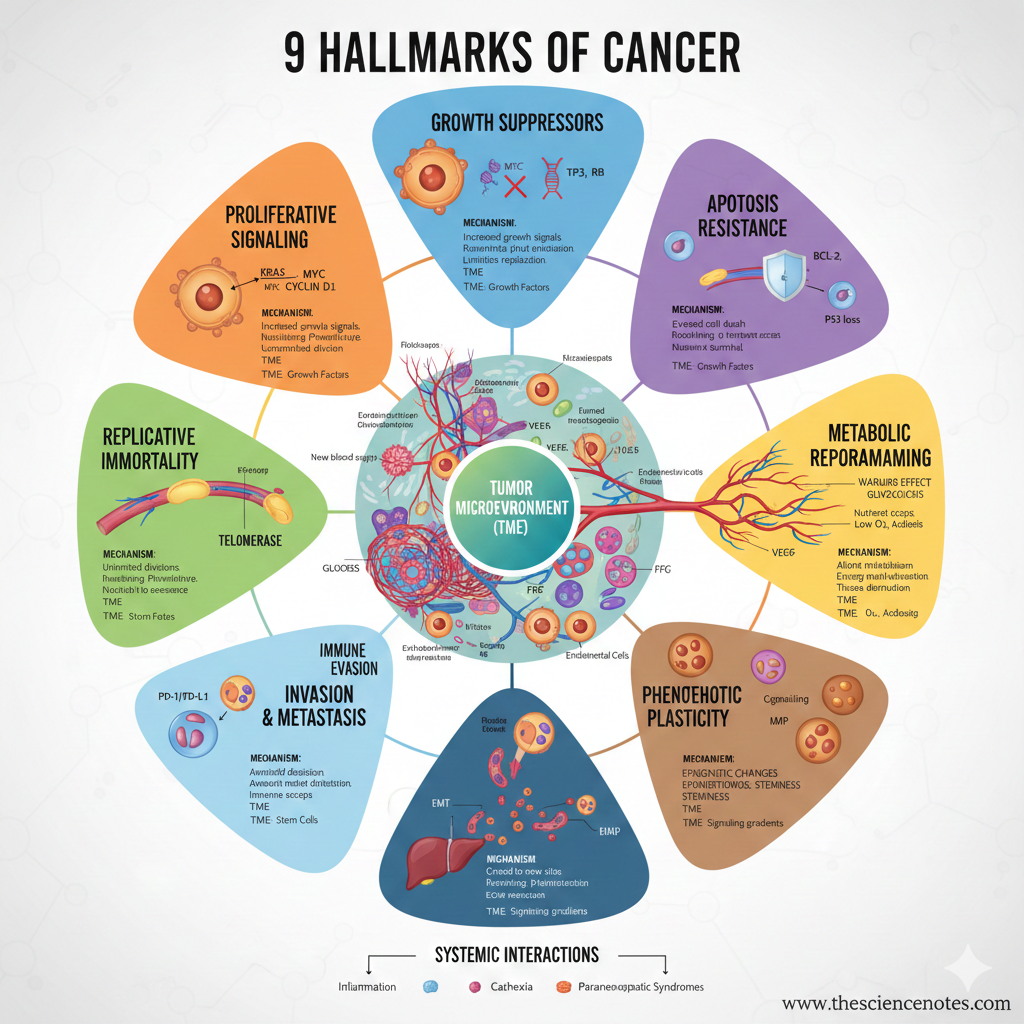
Cancer’s 9 characteristics
1. Maintain proliferative signaling
Cancer cells achieve uncontrolled spread by activating oncogeneswhich drives chronic cell cycle progression.
Important oncogenes:
-
CRAS, NRAS, HRAS: Mutations in KRAS are seen in ~30% of tumors, including pancreatic (~90%), colorectal (~50%) and lung cancers (~35%).
-
BRAF, PIK3CA, BCR-ABL: Drive deviant growth signals.
-
MYC: Transcription factor that regulates thousands of genes; amplified in ~40% of tumors.
Mechanisms:
-
Gene amplification or rearrangement
-
Circular extrachromosomal DNA (ecDNA) increases oncogene expression
-
Epigenetic reprogrammingincluding autocrine/paracrine growth factor loops
Clinical relevance: Targeted therapies include BRAF inhibitors (vemurafenib) and HER2 inhibitors (trastuzumab).
Additional Insights: RAS and MYC can stimulate several other characteristic featuresincluding metabolic reprogramming and angiogenesis, highlighting their centrality in tumor development.
2. Avoid growth inhibitors
Normal cells regulate proliferation via tumor suppressor genes (TSG)which works as gatekeepers for cell cycle checkpoints.
Important TSGs:
-
TP53: Activated in response to DNA damage, oncogenic stress or hypoxia; regulates apoptosis, senescence and cell cycle arrest. Mutated in ~40% of cancers.
-
RB, CDKN1A/B (p21/p27), CDKN2A (p16INK4a/p14ARF): Block progression through G1/S and G2/M transitions.
-
APC: Breaks down β-catenin to prevent uncontrolled proliferation.
Mechanisms of evasion:
Clinical relevance: Drugs like CDK inhibitors (palbociclib, ribociclib) can restore growth inhibition in tumors with TSG inactivation.
3. Resist programmed cell death (apoptosis)
Cancer cells escape cellular suicide mechanismswhich enable survival despite DNA damage or abnormal signaling.
Mechanisms:
-
Overexpression of anti-apoptotic proteins: BCL-2, BCL-XL, MCL-1
-
TP53 inactivation, prevents the induction of pro-apoptotic genes such as PUMA and NOXA
-
Dysregulation of alternative cell death pathways: necroptosis, ferroptosis, pyroptosis, autophagy
Therapeutic: BH3 mimetics (Venetoclax) restore apoptosis i CLL and AMLwith ongoing trials for other forms of cancer.
Paradoxical insight: Apoptotic cells can stimulate tumor-promoting signals in neighboring cells or escape death with partially damaged genomes, which contribute to genomic instability and tumor progression.
4. Establishment of replicative immortality
Normal cells are limited by a mitotic clock dictated by telomere length. Cancer cells bypass this share indefinitely.
Mechanisms:
-
Telomerase activation (TERT): Adds telomere repeats, common in glioblastoma (~80%), melanoma (~60%), bladder cancer (~80%).
-
Alternative lengthening of telomeres (ALT): Recombination-based telomere lengthening, common in mesenchymal and neuroepithelial tumors.
Consequences:
5. Induce or gain access to vasculature (angiogenesis)
Tumors require oxygen and nutrients to grow beyond 1–2 mm.
Mechanisms:
-
Hypoxia triggers VEGFA, ANGPT2 and FGF secretion
-
Endothelial cell activation, sprouting and capillary formation
-
Recruitment of pericytes for vessel stabilization
Tumor vasculature:
-
Leaky, chaotic and poorly perfused
-
Inhibits immune cell infiltration, contributes to immune evasion
Alternative mechanism: Vascular co-opwhere tumors hijack pre-existing vessels, especially after anti-angiogenic therapy.
Clinical relevance: Anti-angiogenic therapy (eg bevacizumab) targets VEGF pathways.
6. Deregulation of cellular metabolism
Cancer cells reprogram their metabolism to meet energy and biosynthetic requirements.
Metabolic strategies:
-
Aerobic glycolysis (Warburg effect) beside oxidative phosphorylation
-
Use of alternative fuels: lactate, glutamine
-
Metabolic cross talk with tumor microenvironment cells (fibroblasts, macrophages, T cells)
TME factors:
-
Hypoxia, acidosis, nutrient gradients
-
Paracrine secretion of metabolites
-
Dynamic adaptations during tumor progression and metastasis
Therapeutic Implications: Targeting glycolysis or glutamine metabolism can disrupt tumor growth.
7. Activation of invasion and metastasis
Cancer cells acquire the ability to spread beyond their origin.
Mechanisms:
-
Epithelial-to-mesenchymal transition (EMT) → motility
-
Extracellular matrix remodeling via MMPs
-
Entry into blood or lymphatic circulation
-
Colonization of distant organs
TME contribution: Stromal cells and immune cells secrete factors that facilitate invasion.
Clinical Significance: Metastases cause ~90% of cancer-related deaths.
8. Avoid immune destruction
Tumors escape immune surveillance via:
-
Immune checkpoints (PD-L1, CTLA-4)
-
Immunosuppressive cytokines
-
Remodeling tumor vasculature to prevent T cell infiltration
Therapeutic methods: Checkpoint inhibitors (nivolumab, pembrolizumab) restore T-cell activity.
9. Unlocking phenotypic plasticity
Cancer cells dynamic switching between proliferative, invasive and drug-resistant states.
Importance:
-
Driver therapy resistance, relapse and metastases
-
Allows adaptation to fluctuating TME and systemic pressure
Activates phenotypic traits
These support the acquisition of characteristics:
-
Genomic instability: accelerates mutations and tumor development
-
Tumor-promoting inflammation: certain immune cells facilitate its growth
-
Epigenetic remodeling and adaptation to oxidative stress increase survival under adverse conditions
Tumor microenvironment (TME)
Tumors are heterogeneous “organs” with several interacting cell types:
-
Cancer cells: spread and adapt
-
Fibroblasts: remodeling the ECM and secreting growth factors
-
Immune cells: suppress or promote tumor growth
-
Blood vessels: deliver nutrients and remove waste
Role: TME contributes to growth, invasion, angiogenesis, immune evasion, metabolic adaptation and therapy resistance.
Systemic interactions
Tumors interact systemically with the body and change:
-
Hormone levels
-
Metabolic balance
-
Immune surveillance
Impact: Systemic effects influence tumor progression and therapy response.
Therapeutic implications
Effective cancer treatment often requires multi-characteristic targeting:
-
Oncogene inhibitors: CRASH, BRAF, HERE2
-
Tumor suppressor modulators: CDK inhibitors
-
Apoptosis activators: BH3 mimetics
-
Anti-angiogenic drugs: VEGF inhibitors
-
Immunotherapy: checkpoint inhibitors
-
Metabolic inhibitors: glycolysis/glutamine targeting
Combination therapy is essential to overcome plasticity, adaptation and resistance.
-
Hanahan, D., & Weinberg, RA (2011). Hallmarks of cancer: The next generation. Cell, 144(5), 646–674. https://doi.org/10.1016/j.cell.2011.02.013
-
Hanahan, D. (2022). Characteristics of cancer: New dimensions. Cancer Discovery, 12(1), 31–46. https://doi.org/10.1158/2159-8290.CD-21-1059
-
Hanahan, D. (2026). Characteristics of cancer – then and now, and beyond. Cell. https://doi.org/10.1016/j.cell.2025.12.049






