Author: Alisha G C
Abstract
Blinatumomab is a first in class bispecific T-cell engager (BiTE) antibody which has fundamentally reshaped the therapeutic landscape to relapsed or refractory B-cell acute lymphoblastic leukemia (B-ALL). By physically connecting endogenous cytotoxic T cells CD19-expressing malignant B cellsBlinatumomab enables potent, major histocompatibility complex (MHC)-independent T-cell cytotoxicitythereby overcoming critical limitations of conventional chemotherapy and antigen presentation-dependent immunotherapies. Despite impressive clinical efficacy, especially in minimal residual disease (MRD)-positive and heavily pretreated patients, therapeutic resistance and disease relapse significant obstacles to lasting remission remain. This comprehensive review provides a thorough analysis of Blinatumomab molecular architecture, immunological mechanism of action, pharmacokinetics, clinical outcomes, resistance pathways and new strategies to overcome therapeutic failurewhich integrates insights from structural biology, immunology and translational research.
Molecular Structure of Blinatumomab
Blinatumomab is a recombinant fusion protein consisting of two single-chain variable fragments (scFvs) derived from targeting monoclonal antibodies CD19 and CD3εrespectively. Unlike conventional monoclonal antibodies, BiTE molecules are compact, flexible and engineered to bring immune effector cells into direct contact with tumor cells.
CD19 scFv: the B-cell targeting domain
CD19 is one 95 kDa type I transmembrane glycoprotein which belongs to the immunoglobulin superfamily and is expressed throughout most stages of B cell development, from pre-B cells to mature B lymphocytes. Functionally, CD19 acts as a coreceptor of the B-cell receptor (BCR) complexwhere it enhances signaling by lowering activation thresholds and regulating downstream pathways such as PI3K–AKT and SYK signaling.
From a therapeutic perspective, CD19 is an ideal immunotherapy target because of:
-
Expression i >95% of B-ALL cases
-
Stable surface localization during leukemogenesis
-
Minimal expression outside the B-cell line, reduces off-target toxicity
The CD19 scFv in Blinatumomab recognizes an extracellular epitope that remains accessible even in conditions of low antigen density, enabling efficient engagement of malignant B cells.
CD3ε scFv: The T-Cell Engagement Domain
CD3ε is an important signaling component in T-cell receptor (TCR) complexassociated with the CD3γ, CD3δ and CD3ζ homodimer. When switched on, CD3ε transduces activation signals through immune receptor tyrosine-based activation motifs (ITAMs) found on CD3ζ chains.
Importantly, Blinatumomab binds CD3ε regardless of antigen specificitywhich allows the recruitment of polyclonal, non-tumor-specific T cells. This mechanism bypasses the need for peptide antigen processing and presentation, a frequent immune evasion strategy in leukemia.
Linker design and structural configuration
The two scFvs are linked via a flexible, non-immunogenic (Gly4Ser)3 linkerproduces one ~55 kDa BiTE molecule. This linker provides sufficient rotational freedom to allow simultaneous binding of CD19 and CD3 without steric hindrance.
A defining feature of Blinatumomab is absence of an Fc domainseam:
-
Improves tissue penetration and diffusion
-
Eliminates Fcγ receptor binding
-
Reduces off-target immune activation, including ADCC and CDC
-
Minimizes non-specific cytokine release
Important structural features
-
Monovalent scFv binding enables rapid serial engagement of multiple tumor cells
-
Flexible linker architecture supports stable immunological synapse formation
-
Short half-life in serum (~2 hours) allows precise pharmacokinetic control via continuous IV infusion
-
Fc-less design improves safety and specificity
Crystallographic and biophysical studies confirm that the spatial orientation of the CD19 and CD3 binding arms is optimal for intercellular tetheringwhich results in the effective formation of a functional cytolytic immunological synapse.
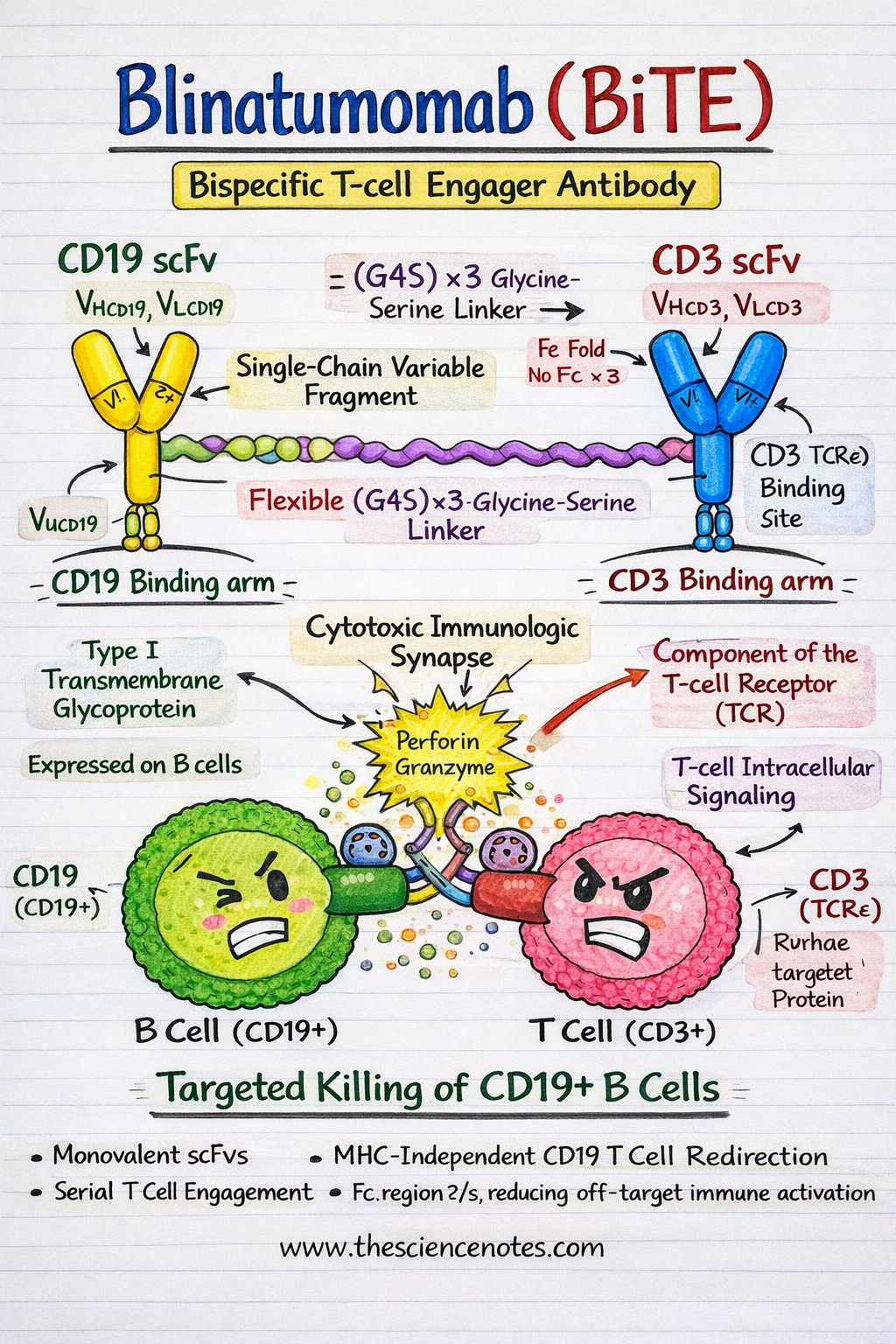
Mechanism of action for Blinatumomab
1. Immunological synapse formation and redirection of T cells
Blinatumomab mediates MHC-independent T-cell cytotoxicity by physical bridge construction CD3+ T cells and CD19+ leukemic B cellswhich efficiently convert resting T cells into serial tumor killers.
-
Two-fold commitment induce TCR clustering through CD3ε while anchoring malignant B cells via CD19
-
This forced proximity mimics physiological immune synapse formation
At the synapse, key signaling molecules such as Lck, LAT, SLP-76, PKCθand actin regulatory proteins are recruited and spatially organized.
Intracellular signal transduction
In case of CD3 involvement:
-
Lck phosphorylates ITAMs on CD3ζ chains
-
ZAP-70 is recruited and activated
-
Adapter proteins (LAT, SLP-76) assemble signaling complexes
-
Calcium influx and diacylglycerol (DAG) production activation:
-
Calcineurin → NFAT
-
PKCθ → NF-KB
-
MAPK cascade → AP-1
-
These transcriptional programs drive T cell activation, proliferation and cytotoxic function.
Effector functions
-
Perforin-granzyme B-mediated apoptosis
-
Release of pro-inflammatory cytokines (IFN-γ, IL-2, TNF-α)
-
Serial murderwhere a single T cell disconnects and targets multiple leukemia cells
2. T cell reprogramming and immune memory
Beyond immediate cytotoxicity, Blinatumomab induces functional reprogramming of T cellspromote expansion of:
These populations contribute to long-term immune monitoring and may underlie persistent MRD negativity observed in responding patients.
3. Pharmacokinetics and immunodynamics
Due to its small size and lack of Fc region, Blinatumomab shows:
-
Quick clearance via renal filtration and proteolysis
-
Peak T-cell activation and expansion between day 7–14
-
Preferably expansion of CD8+ cytotoxic T cellscorrelates with the depth of the response
Continuous intravenous infusion ensures stable plasma concentrations while allowing rapid discontinuation in case of toxicity.
Clinical effect of Blinatumomab in B-ALL
TOWER Trial (NEJM, 2017)
-
Adult relapsed/refractory B-ALL
-
CR/CRh: 43% vs. 25% (Blinatumomab vs chemotherapy)
-
Median overall survival: 7.7 versus 4.0 months
BLAST trial (Blood, 2018)
-
MRD-positive B-ALL in haematological remission
-
MRD approval: 78% after one cycle
-
3-year overall survival: 71%
Adverse events
-
Cytokine release syndrome (CRS): early onset, typically steroid-responsive
-
Neurotoxicity: grade ≥3 in ~10%, potentially linked to T-cell trafficking across the blood-brain barrier
Mechanisms of resistance to Blinatumomab
1. Antigen Escape (CD19 loss)
-
Alternative splicing (eg skipping exon 2)
-
CD19 gene mutations or deletions
-
Lineage switch to myeloid phenotype, particularly in MLL-rearranged leukemia
2. T-cell resistance
-
Up-regulation of fatigue markers (PD-1, TIM-3, LAG-3)
-
TOX-dependent epigenetic reprogramming
-
Metabolic insufficiency and mitochondrial dysfunction
3. Immune checkpoint and microenvironment suppression
-
PD-L1 upregulation on leukemia blasts and MDSCs
-
Immunosuppressive cytokines (IL-10, TGF-β)
-
Expansion of regulatory T cells and suppressive monocytes
Strategies to Overcome Blinatumomab Resistance
Dual and multi-specific targeting
Checkpoint inhibition
T-Cell Fitness Enhancement
Next generation BiTE platforms
Conclusion
Blinatumomab represents a paradigm shift in B-ALL immunotherapyenables precise, MHC-independent T-cell redirection with significant clinical benefit across multiple disease settings. While resistance mechanisms such as antigen escape, T-cell dysfunction and immune suppression remain significant obstacles, rational combination therapies, next generation BiTE technique and immune profiling guided strategies offer promising avenues to improve the durability of the response. Continued integration of molecular biology, immunology and clinical innovation will be critical to expanding the therapeutic window of Blinatumomab and achieving long-term cures in B-ALL.
Frequently Asked Questions (FAQ)
Q1. What is Blinatumomab and how does it work?
Blinatumomab is a bispecific T-cell engager (BiTE) antibody that connects CD3-positive T cells to CD19-positive B cells, triggering MHC-independent T-cell-mediated cytotoxicity.
Q2. Why is CD19 an ideal target in B-ALL?
CD19 is expressed in over 95% of cases of B-cell acute lymphoblastic leukemia and is absent in most non-B-lineage tissues, minimizing off-target toxicity.
Q3. What are the main resistance mechanisms to Blinatumomab?
Resistance arises from loss of CD19 antigen, exhaustion of T cells, immune checkpoint upregulation, and suppressive tumor microenvironment factors.
Q4. How does Blinatumomab differ from CAR-T cell therapy?
Unlike CAR-T therapy, Blinatumomab redirects endogenous T cells without genetic modification and allows precise pharmacokinetic control through continuous infusion.
Q5. What Are New Strategies to Overcome Blinatumomab Resistance?
Approaches include dual-target BiTEs, checkpoint inhibitor combinations, cytokine-based T-cell support, and next-generation trispecific engager platforms.