All matter in the universe is composed of atomsthe smallest individual units of elements. Although atoms were once thought to be indivisible, scientists now know that they are made up of even smaller components called subatomic particles. The structure and behavior of atoms explain the physical and chemical properties of all substances, from simple gases to complex living organisms.
Modern chemistry and physics are built on the understanding of atomic structure. By studying atoms, scientists can explain how elements interact, how compounds form and why matter behaves the way it does.
What is an atom?
An atom is the smallest unit of an element that retains that element’s chemical identity. Each atom consists of three primary subatomic particles:
Together these make up the particles mass and electric charge of an atom. While atoms cannot be broken down by ordinary chemical reactions, their internal structure determines how they interact with other atoms.
Each element – such as hydrogen, oxygen or potassium – consists of atoms with a unique internal structure, especially in the number of protons found in the nucleus.
The history of atomic theory
Early ideas: Democritus and Atomos
The concept of atoms dates back to around 450 BCthen the Greek philosopher Democritus proposed that all matter was made of small, indivisible particles. He called these particles atomswhich means “indivisible”. Although his idea was groundbreaking, it lacked experimental evidence and was largely ignored for centuries.
Dalton’s atomic theory
The atomic theory was revived in the beginning 19th century by English scientist John Daltonwhose ideas form the basis of modern atomic theory. Dalton proposed five key postulates:
-
All matter is composed of tiny particles called atoms.
-
All atoms in a given element are identical.
-
Atoms of different elements are different from each other.
-
Atoms of different elements combine in fixed proportions to form compounds.
-
Atoms cannot be created or destroyed in chemical reactions, only rearranged.
While later discoveries refined some of these ideas, Dalton’s theory was crucial in establishing atoms as the basis of matter.
Discover subatomic particles
Dalton believed that atoms were indivisible, but scientific discoveries in the late 1800s and early 1900s proved otherwise.
The discovery of the electron
IN 1897, JJ Thomson discovered electrona negatively charged particle much smaller than an atom. Since atoms are overall electrically neutral, this discovery raised an important question: how can negative particles exist inside a neutral atom?
Thomson suggested plum pudding modelwhich suggested that electrons were embedded in a positively charged “pudding.” Although incorrect, this model marked the first attempt to describe atomic structure.
Rutherford’s nuclear model
Just a few years later, Ernest Rutherford performed experiments that dramatically changed the understanding of atomic structure. His gold foil experiment showed that:
-
Most of an atom’s mass is concentrated in a small center core
-
The core carries one positive charge
-
Electrons occupy the space around the nucleus
This discovery disproved the plum pudding model and revealed that atoms are most everything empty space.
The discovery of the neutron
IN 1932, James Chadwick discovered neutrona neutral particle found in the nucleus. This discovery completed the basic picture of atomic structure, and explained how atoms could have extra mass at no extra cost.
The structure of an atom
Atoms consist of a central core surrounded by electrons moving in a cloud-like region. Each subatomic particle has distinct properties and functions.
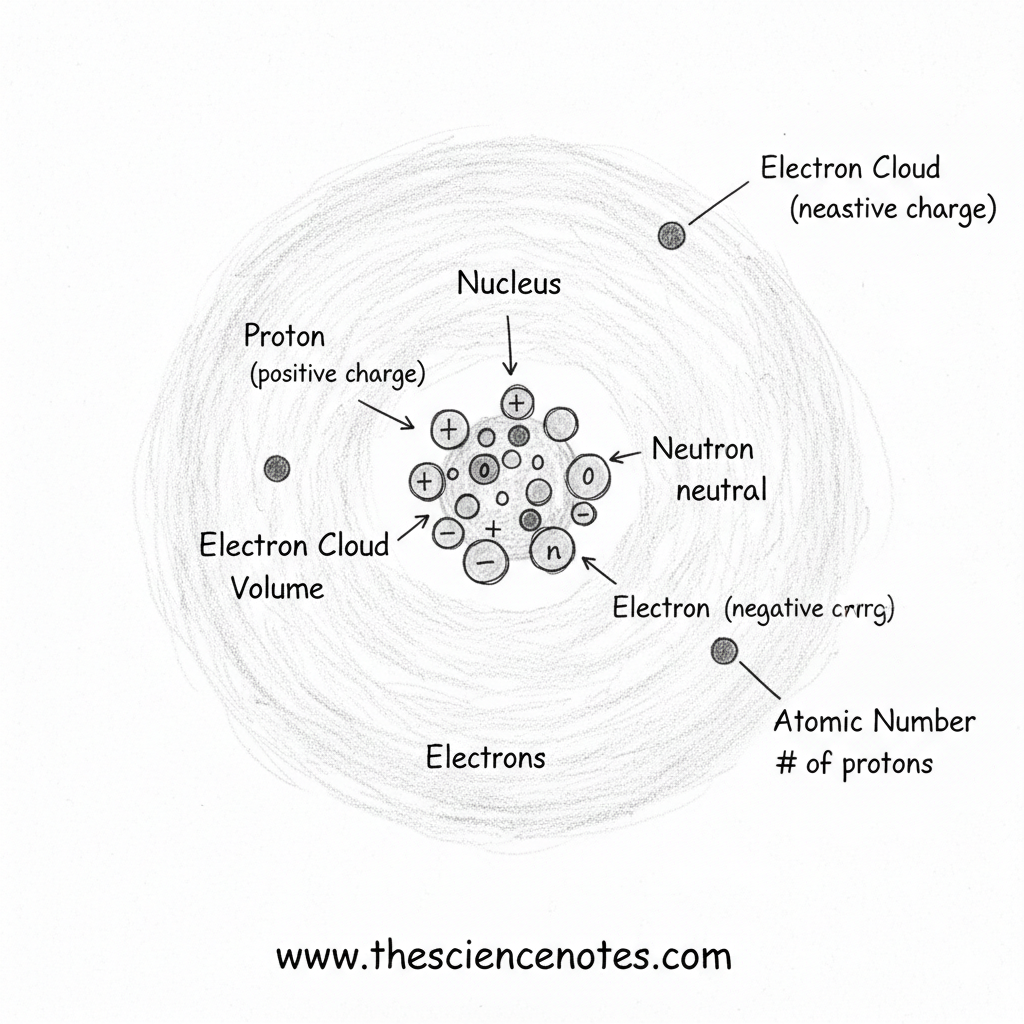
Protons: Define the element
Protons are positively charged particles located in the nucleus of an atom. Each proton has a mass on it one atomic mass unit (AMU).
The number of protons in an atom is known as its atomic numberwhich determines the identity of the element. For example:
-
Hydrogen has 1 proton
-
Carbon has 6 protons
-
Oxygen has 8 protons
Changing the number of protons changes the element itself.
Neutrons: Adds mass and stability
Neutrons is also found in the nucleus and has no electrical charge. As protons, they have a lot of mass an AMUwhich contributes significantly to the total mass of the atom.
Neutrons play an important role in stabilizing the nucleus. Atoms of the same element can have different numbers of neutrons that are formed isotopes with slightly different atomic masses.
Electrons: The cloud around the nucleus
Electrons are negatively charged particles that move rapidly around the nucleus in regions known as electron clouds. These clouds account for most of an atom volumeeven though electrons have negligible mass.
Electrons remain near the nucleus due to their attraction to the positively charged protons. Their arrangement, known as electron configurationdetermines how atoms interact and bond with each other.
Atomic mass and charge
The atomic mass of an atom is determined primarily by the combined mass of its protons and neutrons. Electrons contribute very little to the mass, but play a decisive role in the charge balance.
In a neutral atom:
This balance gives the atom no net electric charge.
Ions and electron configuration
Atoms are not always electrically neutral. If an atom gains or loses electrons, it becomes a ion.
Ion formation is critical in chemical reactions, biological processes and electrical conductivity.
Why atomic structure is important
Understanding atomic structure helps explain:
-
Chemical reactions and bonding
-
Physical properties of substances
-
Electrical behavior of materials
-
Biological processes at the molecular level
From the formation of water molecules to the transmission of nerve impulses, atomic interactions shape the natural world.
Conclusion
Atoms are the basic units of matter, composed of protons, neutrons and electrons. Centuries of scientific discovery—from Democritus to Dalton to modern physicists—have revealed the intricate structure hidden within these tiny particles.
By understanding atomic structure, scientists can explain the behavior of elements, predict chemical reactions and unlock new technologies. Although atoms are incredibly small, their impact on the universe is enormous.






