Bacterial transformation is a cornerstone technique in molecular biology, biotechnology and genetic engineering. That allows the researchers to introduce foreign DNA into bacterial cells, making DNA possible cloning, gene expressionand large scale protein production. Among the various transformation techniques available are heat shock method is still one of the most widely used due to its simplicity, reasonable prices and reliability – especially when working with Escherichia coli (E.coli).
This article provides one detailed, step-by-step educational overview of bacterial transformation using heat shock technique. That explains biological principle, laboratory procedure, applicationsand downstream usewhich makes it ideal for students, candidates and early career researchers.
What is bacterial transformation?
Transformation is the process that a bacterial cell takes up extracellular DNA from their environment. This DNA can then be maintained, replicated or expressed inside the cell.
In nature are some bacterial species naturally competentwhich means they can spontaneously absorb DNA. However, there are many commonly used laboratory strains – such as E.coli– demand artificial induction of competence.
Cells that are able to take up DNA are called competent cells. In molecular biology laboratories, competence is typically induced through the use of chemical or physical methods.
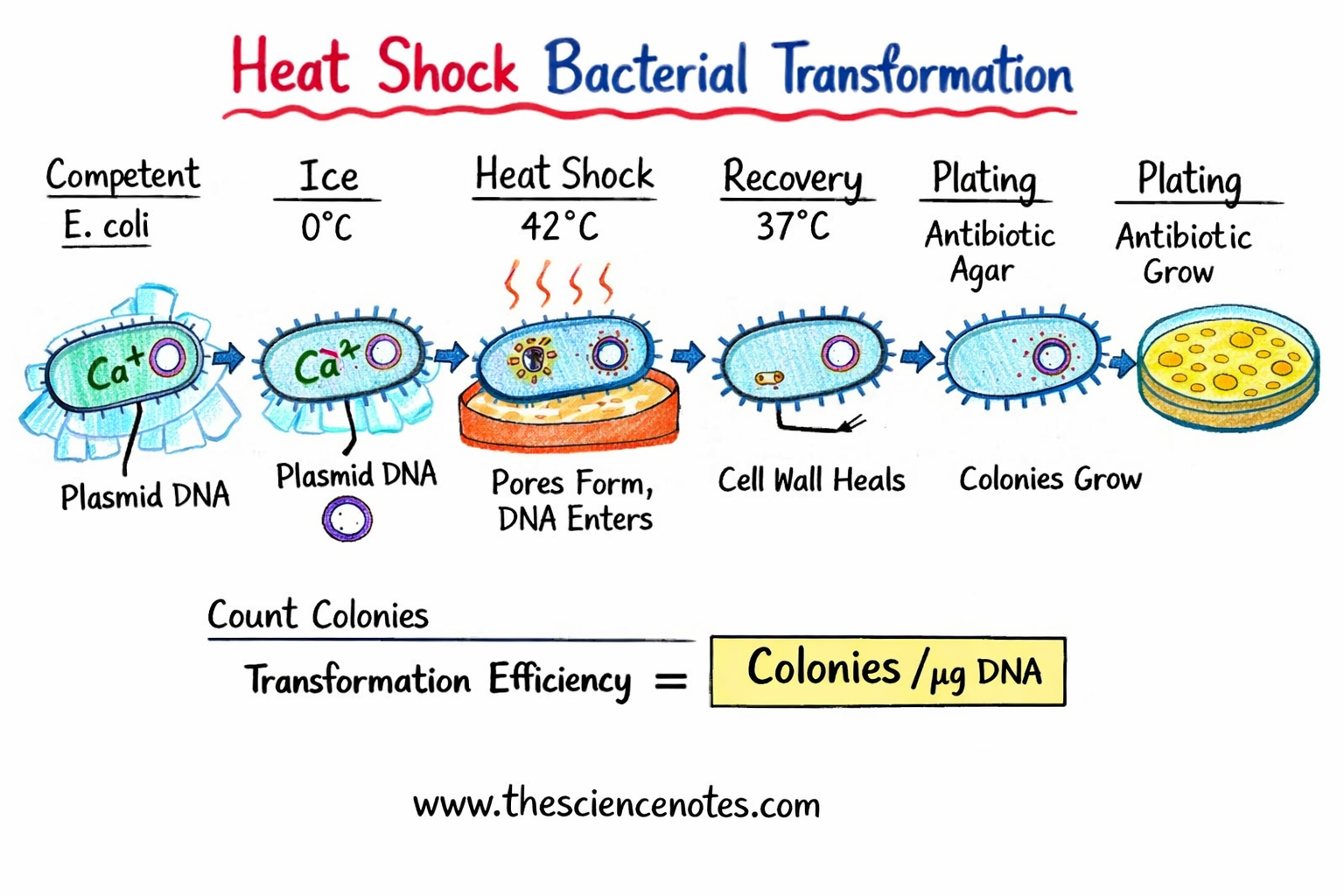
What is heat shock transformation?
Heat shock transformation is an artificially induced method that temporarily increases the permeability of the bacterial cell membrane, allowing plasmid DNA to enter the cell.
Core principle
-
Bacterial cells are exposed to a calcium-rich environmentusually calcium chloride (CaCl2).
-
Calcium ions neutralize negative charges on both plasmid DNA and the bacterial cell surface.
-
A brief exposure to high temperature (42°C) creates a thermal and pressure imbalance over the membrane.
-
This imbalance leads to the formation of temporary poresthrough which plasmid DNA enters.
-
When the cells are returned to cold or physiological temperature, the membrane is resealed.
Plasmids: The DNA molecules used in transformation
The most commonly used DNA in bacterial transformation experiments is plasmid.
What is a plasmid?
A plasmid is a small, circular, double-stranded DNA molecule which exist independently of the bacterial chromosome. Plasmids can supercoil, which reduces their size and makes them more likely to pass through membrane pores during transformation.
Important properties of a plasmid
1. Multiple Cloning Site (MCS)
The multiple cloning sites contains short DNA sequences recognized by restriction endonucleases. These enzymes cut DNA at specific locations, allowing researchers to insert a gene or DNA fragment of interest into the plasmid.
2. Origin of Replication (ORI)
The origin of replication tells the bacterial cell where to start copying the plasmid. Without a functional ORI, the plasmid cannot be maintained inside the cell.
3. Antibiotic resistance gene
Most plasmids include a gene that confers resistance to a specific antibiotic (such as ampicillin or kanamycin). This function activates selectionwhich ensures that only bacteria containing the plasmid survive on antibiotic-containing media.
Competent cells: Prepare bacteria for DNA uptake
Why E.coli Is often used
E.coli is the preferred organism for transformation because it:
-
Grows quickly
-
Is genetically well characterized
-
Is easy to culture and manipulate
-
Gives high plasmid yields
Chemical competence when using calcium chloride
Competence is induced by exposing bacterial cells to cold calcium chloride:
-
Calcium ions shield negative charges on DNA and the bacterial membrane
-
Electrostatic repulsion is reduced
-
The cell wall becomes more permissive for DNA penetration
Cells are kept on ice throughout this process to stabilize the membrane before heat shock.
Mechanism of heat shock transformation
The transformation process involves several coordinated steps:
-
DNA binding
Plasmid DNA associates with the bacterial surface during cold incubation. -
Thermal shock
A rapid change to 42°C creates a pressure difference across the membrane. -
Pore formation
Temporary pores form in the membrane. -
DNA introduction
Supercoiled plasmid DNA enters the cytoplasm. -
Membrane restoration
Cooling allows the membrane to reseal, trapping the DNA inside.
Step-by-step heat shock transformation protocol
Preparation and sterility
-
Clean the work area thoroughly
-
Sterilize all solutions and instruments
-
Prepare LB media and LB agar plates
-
Add antibiotics to agar at 50–55°C
-
Allow the plates to harden at room temperature
-
Preheat the antibiotic plates to 37°C
-
Set the water bath to 42°C
Transformation procedure
-
Melt chemically competent E.coli cells on ice
-
Add 1–5 µL of cold plasmid DNA (≈1 ng/µL)
-
Mix gently and incubate on ice for 30 minutes
-
Heat shock cells at 42°C for 30 seconds
-
Return the tubes to ice immediately
-
Add 450 µL recovery medium
-
Incubate at 37°C for 1 hour with shaking (>225 rpm)
-
Plate 20–200 µL of transformed cells on LB agar with antibiotics
-
Incubate the plates overnight at 37°Creverse
Selection and identification of transformants
After overnight incubation, bacterial colonies appear on antibiotic plates.
Why antibiotic choice works
Only cells that have taken up the plasmid successfully – and therefore express antibiotic resistance gene– can grow. Non-transformed cells are eliminated.
Calculation of transformation efficiency
Transformation efficiency measures how effective the transformation was.
Formula:
Transformation efficiency =
(Number of colonies × dilution factor) ÷ DNA coated (µg)
This calculation is essential for comparing different protocols, plasmids or competent cell batches.
Preparation of chemically competent cells (Overview)
-
Grow bacteria to in the middle of the log phase (measured by optical density)
-
Chill the cells on ice to stop growth
-
Centrifuge at 4 °C and discard the supernatant
-
Wash the cells several times with cold 0.1 M CaCl 2
-
Finally re-hanging in CaCl2 + 15% glycerol
-
Aliquots in microcentrifuge tubes
-
Shop at -80°C
Alternative transformation methods
Electroporation
Electroporation uses a card electrical pulse to create membrane pores. It provides higher efficiency, but requires specialized equipment and salt-free DNA preparations.
Blue-white screening
Many plasmids contain lacZ gene that codes for β-galactosidase:
This method facilitates screening for recombinant clones.
Applications of bacterial transformation
Bacterial transformation enables:
-
DNA cloning and plasmid amplification
-
Gene expression studies
-
Recombinant protein production
-
Functional genomics
-
Structural biology and crystallography
Downstream applications after transformation
Plasmid purification
Transformed bacteria are grown in liquid media with antibiotics. Plasmids are isolated using commercial purification kits.
Protein expression and purification
In expression experiments:
-
Bacteria produce large amounts of protein
-
Cells are lysed
-
Target proteins are purified using affinity chromatography
-
Proteins can be crystallized for structural analysis
Conclusion
The heat shock method of bacterial transformation is a fundamental technique that underpins modern molecular biology. By combining chemically competent cells, calcium chlorideand a card thermal shockresearchers can effectively introduce plasmid DNA into bacteria.