Elements are the basic building blocks of matter. They are the smallest units that cannot be broken down into simpler substances through chemical processes. Everything around us – air, water, soil and living organisms – is made up of combinations of these elements. Although researchers have identified 118 known elementsonly a fraction are naturally occurring, and even fewer are essential for life.
Understanding how elements are organized, how they behave and how they contribute to living systems is crucial for fields such as chemistry, biology, medicine and environmental science. At the heart of this understanding lies periodic tablea powerful tool that reveals the physical and chemical properties of all known elements.
What are elements and why are they important?
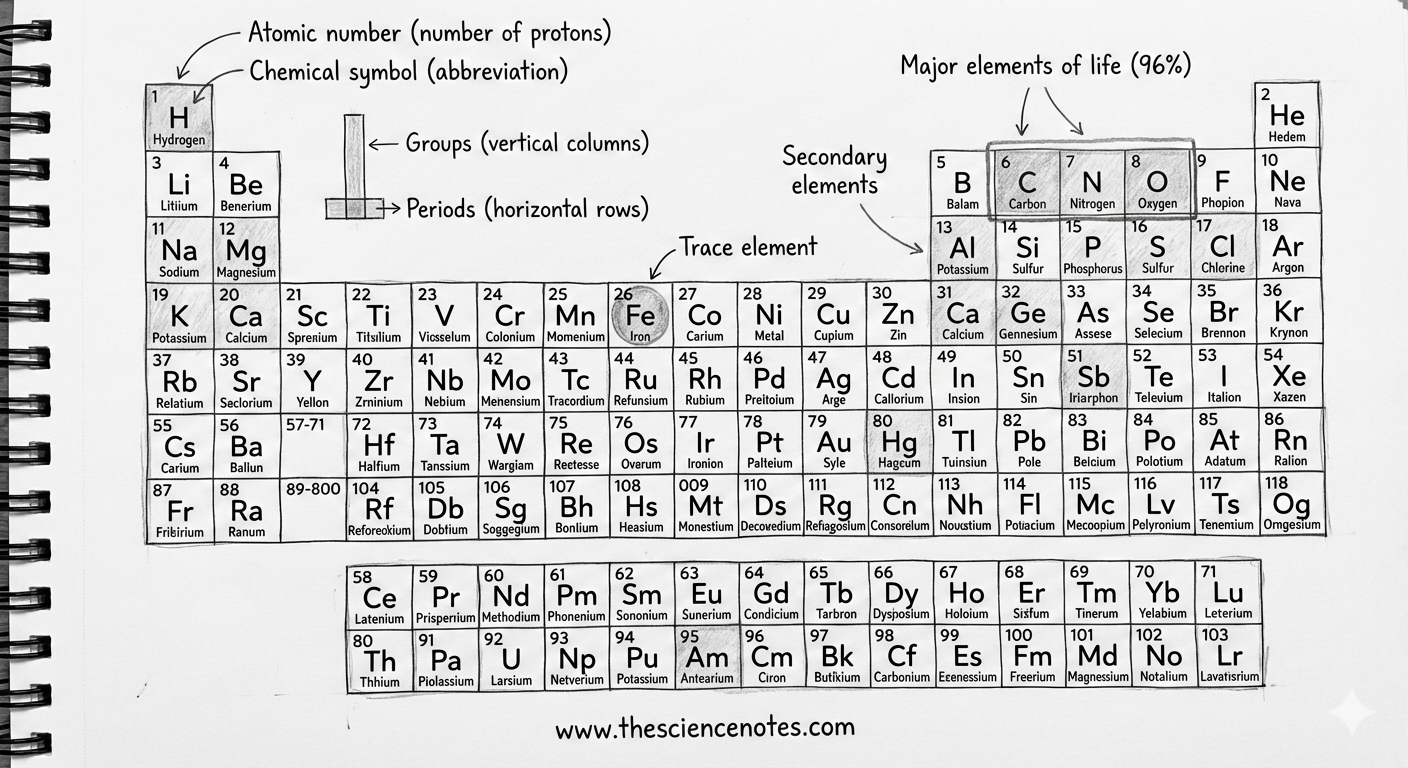
An element is a pure substance consisting of only one type of atom. Each element is defined by its own atomic numberwhich represents the number of protons in the nucleus. This number determines an element’s identity and its position in the periodic table.
While there are 118 known elements, only 92 occur naturally on earth. Even more striking is the fact that humans require only 25 items to live and reproduce. Only of these four elements – oxygen, carbon, hydrogen and nitrogen – make up about 96% of all living matter.
These elements are essential because they form the molecules that support life, including proteins, carbohydrates, lipids and nucleic acids.
The periodic table: Organization of the elements
The periodic table is a systematic arrangement of all known elements based on increasing atomic number and recurring chemical properties. Each square on the table provides important information, including:
-
Atomic number
-
Chemical symbol
-
Element name
-
Atomic weight
For example, sodium has the chemical symbol Nowderived from the Latin name sodium. This standardized system allows researchers worldwide to communicate clearly and consistently.
Groups and periods: Patterns in chemical behavior
Beyond atomic numbers, the periodic table is organized into columns (groups) and rows (period) which reflect deeper chemical relationships.
Groups (columns)
Elements in the same group share similarities chemical propertiesalthough their atomic sizes may vary. This similarity arises because they have the same number of electrons in their outermost shell, which largely determines how elements react and bond with others.
Periods (rows)
Elements in the same period are more similar in size and have their electrons arranged in comparable energy levels. However, their chemical properties can vary widely over a period when the number of protons and electrons increases.
This unique organization allows scientists to predict how elements will behave, even if they have never been studied before.
Main elements that make up the human body
All living organisms on earth contain oxygen, carbon, hydrogen and nitrogen. In humans, these four elements stand for 96% of total body mass.
-
Oxygen (O): Necessary for cellular respiration and energy production
-
Carbon (C): The backbone of all organic molecules
-
Hydrogen (H): Plays a key role in energy transfer and molecular structure
-
Nitrogen (N): Crucial for proteins and nucleic acids such as DNA
These elements form complex molecules that drive every biological process, from metabolism to reproduction.
Secondary elements in living matter
The remaining ones 4% of the human body mainly consists of the following elements, listed in order:
-
Calcium (Ca): Supports bones, teeth and muscle function
-
Phosphorus (P): Important for DNA, RNA and energy transfer (ATP)
-
Potassium (K): Regulates nerve signals and fluid balance
-
Sulfur (S): Found in certain amino acids and proteins
-
Sodium (Na): Contributes to maintaining fluid balance and nerve impulses
-
Chlorine (Cl): Important for digestion and electrolyte balance
-
Magnesium (Mg): Necessary for enzyme function and muscle activity
Although present in smaller amounts, these elements are essential for maintaining normal physiological functions.
Trace elements: small amounts, big impact
Track elements are elements required by the body in extremely small amounts – less than 0.01% of total body weight– Nevertheless, they are crucial for health and survival.
Iron: A critical trace element
One of the most important trace elements is iron (Fe). Iron plays a central role in red blood cells by helping hemoglobin to bind oxygen and transport it throughout the body.
Lack of iron can lead to iron deficiency anemiaa condition marked with:
-
Exhaustion
-
Shortness of breath
-
Weakness
-
Irregular heartbeat
This highlights how even small amounts of certain elements can have profound effects on human health.
Harmful elements and their effects on living organisms
Not all elements are favorable. Some may be poisonous or even fatalespecially heavy metals.
Toxicity to mercury and heavy metals
Mercury is a heavy metal that can cause serious health problems even at low levels of exposure. Depending on the tissue affected, mercury toxicity can damage the nervous system, kidneys and other organs. In larger doses it can be fatal.
One of the biggest concerns with heavy metals is their ability to do so accumulates in living tissue over timea process known as bioaccumulation.
Environmental impact and bioremediation
Heavy metals often enter ecosystems through industrial pollution and can move through food webbeginning with primary producers such as plants and algae and ultimately affecting higher trophic levels, including humans.
Scientists explore bioremediationa biological approach to removing heavy metal contaminants from the environment. This research requires a deep understanding of both:
Such efforts are essential to protect ecosystems and human health.
Conclusion
Elements are the basis of all matter and life itself. From the four major elements that dominate living matter to trace elements that quietly support essential biological functions, each plays a unique role. The periodic table provides a powerful framework for understanding these elements, their properties, and their interactions.
At the same time, awareness of harmful elements such as mercury emphasizes the importance of responsible environmental management and ongoing scientific research. By studying elements and their behavior, we gain insight not only into the nature of matter, but also into the delicate balance that sustains life on Earth.






