Introduction: Understanding the Cell Cycle and the CFSE T Cell Proliferation Assay
The cell cycle and CFSE T cell proliferation assay is a central theme in modern biology, especially within cell biology, immunology and biomedical research. Every living organism depends on controlled cell division to grow, repair tissues and respond to environmental changes.
The cell cycle refers to the orderly sequence of events that a cell undergoes to duplicate its DNA and divide into two daughter cells. This process occurs throughout an organism’s life and is carefully regulated to maintain cellular health and genetic stability.
Cell division is essential for:
-
Embryonic development from a single fertilized egg (zygote)
-
Growth of tissues and organs
-
Replacement of damaged or dead cells
-
Activation of the immune system
-
Maintenance of organ function
-
Healing of wounds and injuries
In scientific research, it is extremely important to understand how cells divide. A powerful experimental method used to measure immune cell division is CFSE T cell proliferation assaywhich uses fluorescent labeling combined with flow cytometry.
This technique allows researchers to:
-
Track how many times cells divide
-
Measure immune cell activation
-
Study disease progression
-
Evaluate response to treatments
The cell cycle is a highly controlled biological process consisting of several sequential phases. Each phase ensures that the cell is fully prepared before moving on to the next step.
Cells can either remain inactive or actively divide depending on the organism’s physiological needs.
1. Gap 0 (G0) phase – the resting state
The G0 phase is considered a non-dividing stage of the cell cycle.
Important functions of the G0 phase include:
-
Cells are metabolically active but do not divide
-
Cells perform specialized functions
-
Many differentiated cells remain in G0 permanently
-
Some cells can re-enter the cycle when stimulated
Examples of cells often in G0:
This stage helps the body conserve energy and maintain cellular stability.
The G1 phase is the first stage of active cell cycle progression.
Several critical processes occur during G1:
-
The cell increases in size
-
Proteins required for DNA replication are synthesized
-
Cellular organelles multiply
-
The cell evaluates environmental conditions
-
DNA integrity is checked
Important checkpoint events include:
If conditions are unfavorable, the cell may revert to G0 phase instead of continuing the cycle.
3. S phase – DNA synthesis
The S phase is when the most critical event in the cell cycle occurs: DNA replication.
Major events in the S phase include:
-
Duplication of chromosomes
-
Formation of sister chromatids
-
Replication of histone proteins
-
Increased DNA content inside the nucleus
-
Preparation for accurate cell division
At the end of this phase, each chromosome consists of two identical chromatids.
4. Gap 2 (G2) phase – final preparations for division
The G2 phase ensures that the cell is completely ready for mitosis.
Important processes include:
-
Verification of successful DNA replication
-
Repair of DNA damage
-
Synthesis of mitotic proteins
-
Organization of microtubules
-
Preparation of spindle apparatus
This checkpoint prevents damaged or incomplete DNA from being passed on to daughter cells.
5. M phase – mitosis and cytokinesis
The M phase represents the actual division of the cell.
This phase consists of two main processes:
Mitosis
Mitosis includes several stages:
-
Prophase
-
Metaphase
-
Anaphase
-
Telophase
During mitosis:
-
Chromosomes condense
-
The nuclear envelope collapses
-
Chromosomes align in the middle
-
Sister chromatids separate
Cytokinesis
Cytokinesis is the final step in which:
To understand cell cycle and CFSE T cell proliferation assay is extremely important in several scientific disciplines.
In immunology
Cell cycle analysis helps researchers understand immune responses such as:
-
Activation of T cells during infection
-
Expansion of immune cell populations
-
Immune response to vaccines
-
Detection of immune disorders
T cells multiply when they recognize pathogens or abnormal cells.
In cancer biology
Cancer is primarily caused by uncontrolled cell division.
Cell cycle analysis helps researchers:
-
Study tumor growth
-
Identify abnormal scattering patterns
-
Consider cancer treatments
-
Develop targeted therapies
In medical research
Researchers use cell proliferation assays to:
-
Study autoimmune diseases
-
Examine inflammatory responses
-
Assess drug effects
-
Develop new immunotherapies
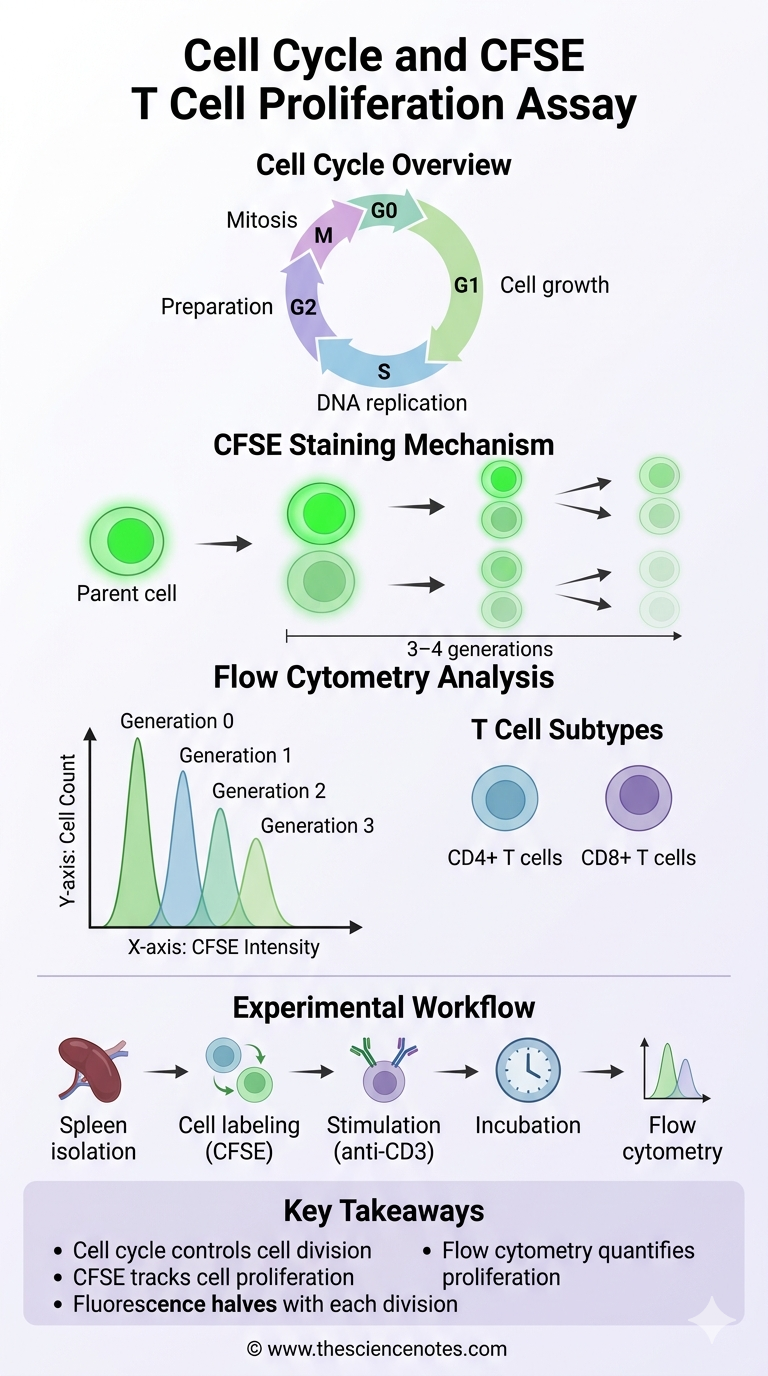
The CFSE T cell proliferation assay is a widely used technique to track cell division in immunological experiments.
CFSE stands for:
Carboxyfluorescein succinimidyl ester
This fluorescent dye allows researchers to observe how cells divide over time.
How CFSE works
The mechanism of CFSE staining involves several important steps:
-
CFSE readily enters living cells.
-
The dye binds permanently to intracellular proteins.
-
All cells initially show the same fluorescence intensity.
-
When a cell divides:
-
Each new generation has half the fluorescence of the previous one.
As a result:
-
First generation cells show the highest fluorescence
-
The second generation shows reduced fluorescence
-
Later generations show progressively lower fluorescence
This pattern forms multiple peaks in flow cytometry analysis.
Flow cytometry is an important analytical technique used in cell cycle and CFSE T cell proliferation assay.
This technology allows rapid analysis of thousands of cells individually.
What flow cytometry measures
Flow cytometry can measure:
-
Cell size
-
Cell complexity
-
Fluorescence intensity
-
Surface protein markers
-
Intracellular proteins
Important flow cytometry parameters
Forward Scatter (FSC)
Indicates cell size.
Side Spread (SSC)
Indicates internal complexity.
Fluorescence channels
Used to detect:
-
CFSE signal
-
CD Markers
-
Other fluorescent labels
1. The preparation stage
Important preparation steps include:
-
Use laboratory gloves and protective clothing
-
Sterilization of dissection instruments
-
Prepare HBSS with fetal calf serum
-
Labeling of test tubes
Proper preparation ensures experimental accuracy and prevents contamination.
2. Mouse spleen dissection
Steps Involved:
-
Euthanize mice using the carbon dioxide system
-
Safe mouse on the dissecting board
-
Perform stomach incision
-
Find the spleen attached to the abdomen
-
Carefully isolate spleen tissue
-
Transfer the spleen to HBSS solution
The spleen is rich in immune cells, making it ideal for proliferation studies.
3. Isolation of immune cells
The aim of this step is to obtain splenocytes.
The procedure includes:
-
Placement of spleen on cell strainer
-
Crushing of tissue to release cells
-
Collect dissociated cells
-
Centrifugation to pellet cells
-
Glowing red blood cells
-
Washing of immune cells
-
Adjustment of final cell concentration
The cells are then ready for staining and stimulation.
Key steps:
-
Divide cells into experimental groups
-
Add CFSE dye
-
Incubate cells
-
Wash off excess dye
-
Stimulate selected samples with anti-CD3 antibody
-
Culture cells in incubator
Two groups are analyzed:
Control group:
Stimulated group:
-
Activation of T cells
-
Increased cell division
Students often struggle with flow cytometry analysis, so here is a simplified overview.
Step 1: Identify lymphocytes
Use FSC vs SSC plot.
Step 2: Gate T cells
Select CD3 positive cells.
Step 3: Identify subsets
Separate:
Step 4: Analyze CFSE signal
Look at histogram peaks that represent cell generations.
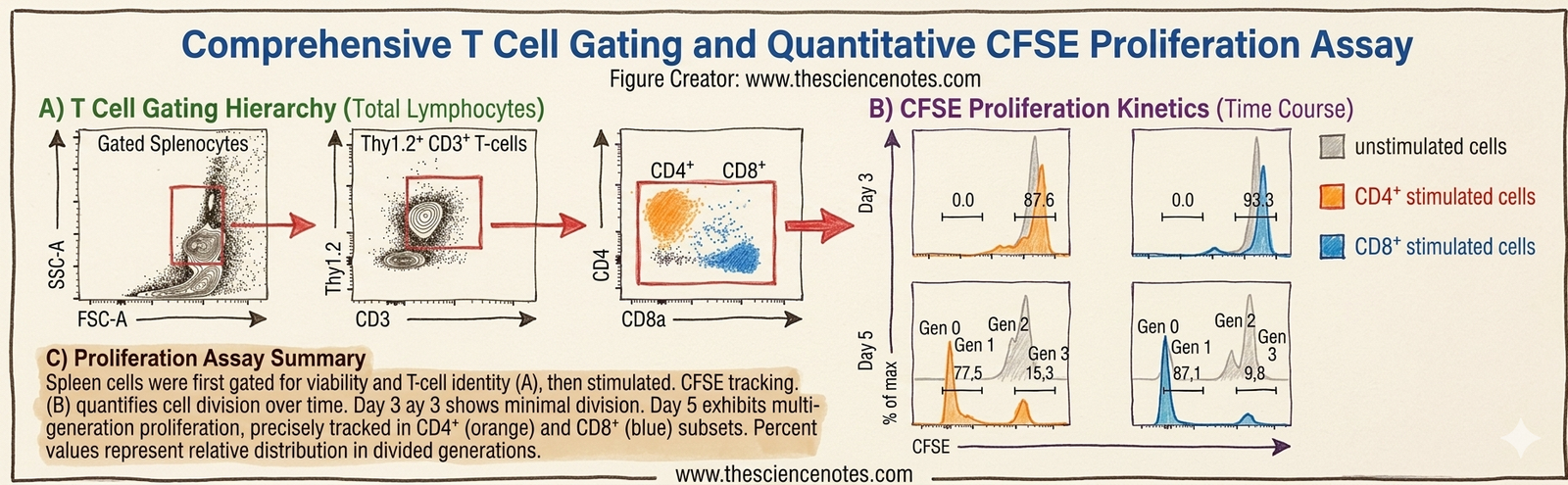
Observations after 3 days
Observations after 5 days
Key experimental findings
-
CD4 T cells proliferate after stimulation
-
CD8 T cells show stronger proliferation
-
Stimulation significantly increases cell division
This analysis is widely used in modern research.
Main applications include:
Other methods include:
BrdU analysis
Labels newly synthesized DNA.
EdU analysis
More sensitive and faster than BrdU.
Fucci mouse model
Allows real-time visualization of cell cycle phases.
To understand cell cycle and CFSE T cell proliferation assay is essential for students studying life sciences.
Key learning points include:
-
Cell cycle phases regulate cell division
-
CFSE staining tracks cell proliferation
-
Flow cytometry provides quantitative analysis
-
Immune cells divide in response to stimulation
-
These techniques are widely used in biomedical research