Proteins are the workhorses of the biological world. From the collagen that gives your skin elasticity to the hemoglobin that transports oxygen through your blood vessels, these macromolecules are essential to every facet of cellular life. Often described as the “building blocks” of the body, proteins are far more complex than simple bricks; they are intricate biological machines whose function is dictated entirely by their form.
In this comprehensive guide, we will explore the hierarchical nature of protein organization—from simple chains of amino acids to complex multi-subunit molecular motors.
What are proteins? A biological overview
At their most basic level, proteins are polymers of amino acid residues. While there are hundreds of amino acids in nature, only 20 serve as standard building blocks for human proteins. These 20 amino acids are arranged in almost infinite combinations to create the great variety of proteins found in living organisms.
The versatility of proteins
Proteins don’t just provide structure; they are dynamic participants in cellular metabolism. Their roles include:
-
Catalysis: Enzymes such as amylase speed up chemical reactions.
-
DNA Replication: DNA polymerase ensures that genetic information is copied accurately.
-
Molecular transport: Membrane proteins move ions and nutrients across cell boundaries.
-
Structural support: Keratin and collagen provide mechanical strength to tissue.
To perform these specific tasks, a protein must fold into a precise three-dimensional shape. This folding process follows a strict organizational hierarchy.
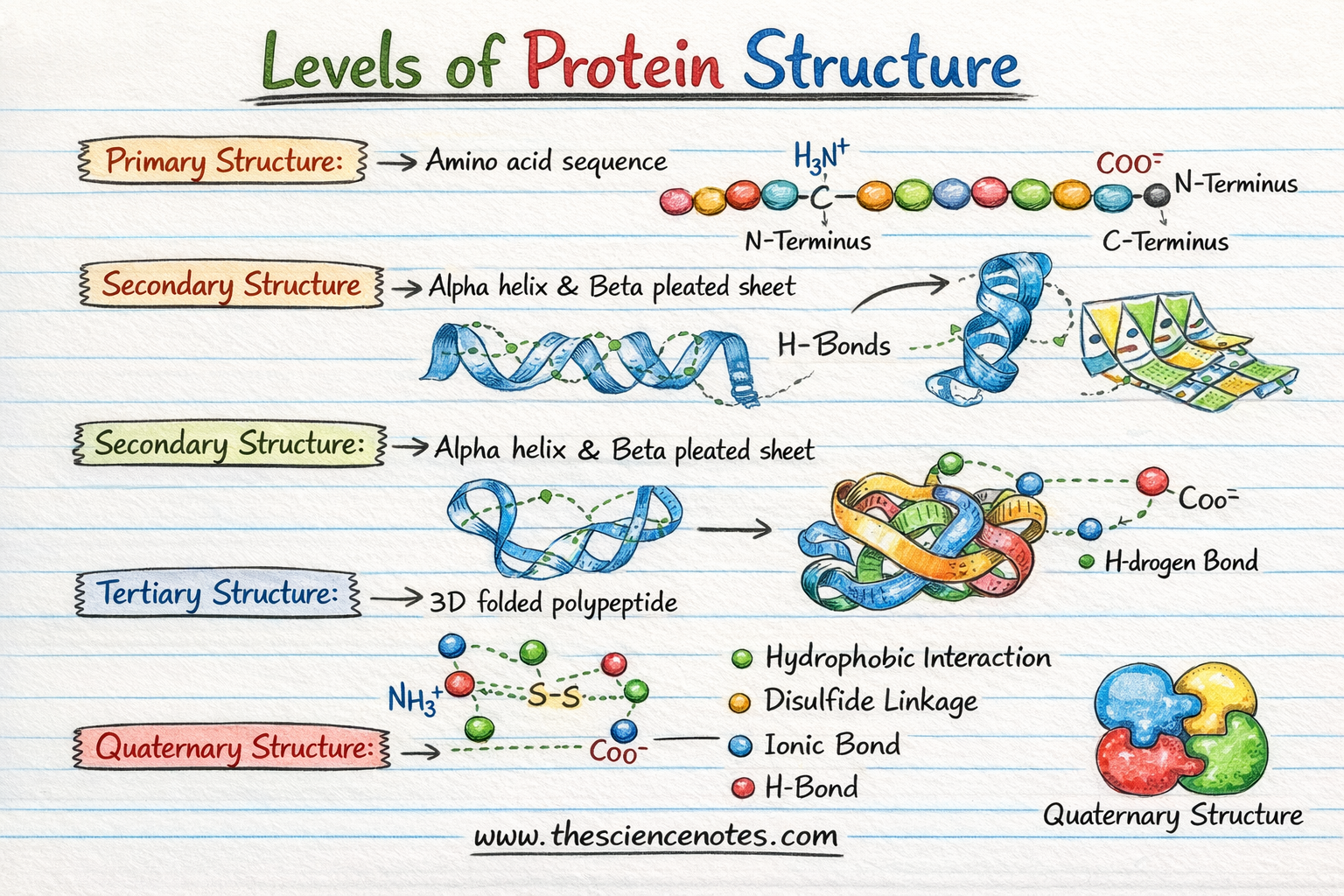
1. Primary structure: The linear blueprint
The primary structure is the simplest level of protein organization. it refers to the unique linear sequence of amino acids in a polypeptide chain.
The peptide bond
Amino acids are linked together by peptide bonds-a type of covalent bond formed between the carboxyl group of one amino acid and the amino group of the next. This creates a “backbone” for the protein.
Direction: N-Terminus to C-Terminus
Each polypeptide chain has a distinct direction:
-
Amino terminal (N-terminal): The end with a free amino group.
-
Carboxyl terminal (C-terminal): The end with a free carboxyl group.
Why it’s important: The primary sequence is determined by the genetic code (DNA). Even a single change in this sequence – such as the mutation found in sickle cell anemia – can completely change the final shape and function of the protein.
2. Secondary structure: Local folding patterns
As the polypeptide chain exits the ribosome during translation, it does not remain a straight line. It begins to fold into localized patterns known as secondary structure. These forms are primarily stabilized by hydrogen bonds between the carbonyl oxygen and the amide hydrogen in the polypeptide backbone.
The Alpha-Helix ($\alpha$-helix)
The $\alpha$-helix is a right-handed spiral. It is held together by hydrogen bonds that form between every fourth amino acid residue. This structure is common in proteins that must be elastic or span cell membranes.
The beta pleated sheet ($\beta$-sheet)
$\beta$-sheet consists of two or more segments of a polypeptide chain lined up side by side. The spine forms a zigzag or “folded” appearance. These segments can be run parallel (in the same direction) or anti-parallel (in opposite directions), creating a very rigid and stable framework.
3. Tertiary structure: The three-dimensional fold
The tertiary structure represents the full geometric form of a single polypeptide chain. This is the level where the protein really assumes its functional form. While the secondary structure is stabilized by the backbone, the tertiary structure is stabilized by interactions between amino acid side chains (R groups).
Forces stabilization of the 3D fold
Several chemical forces work together to “lock” the protein into its original state:
-
Hydrophobic interactions: Non-polar amino acids cluster in the middle of the protein to keep away from water.
-
Hydrogen bonds: Formed between polar side chains.
-
Ionic bonding (salt bridges): Interactions between positively and negatively charged R groups.
-
Disulfide bridges: Strong covalent bonds formed between two cysteine residues. This is the “glue” that makes proteins such as insulin so stable.
-
Van der Waals forces: Weak, short-range attractions that help pack the protein tightly.
4. Quaternary Structure: Multi-Unit Complex
Not all proteins stop at the tertiary level. Some functional proteins consist of two or more polypeptide chains (now called subunits) that work together as a single unit. This is known as quaternary structure.
Homomers vs. heteromeric complexes
Example: Hemoglobin
Hemoglobin, the protein that carries oxygen in our blood, is a classic example of quaternary structure. It consists of four subunits – two alpha-globins and two beta-globins – that must remain bound together to function properly.
Summary table: Protein organization at a glance
| Level | Definition | Stabilizing bonds/forces |
| Primary | Linear sequence of amino acids | Peptide (covalent) bonds |
| Secondary | Local spirals ($\alpha$) or sheet ($\beta$) | Hydrogen bonds (backbone) |
| Tertiary | Full 3D shape of a chain | R-group interactions (disulfide, hydrophobic, etc.) |
| Quaternary | Interaction between multiple chains | Same as tertiary (but between different subunits) |
Conclusion: Form follows function
In biology, the rule “form follows function” is nowhere more evident than in protein folding. The primary sequence dictates the secondary folds, which determine the tertiary 3D shape, which allows the protein to anchor with specific molecules like a key in a lock. When a protein loses this structure – a process called denaturation– It loses its ability to function, which can lead to diseases such as Alzheimer’s or Parkinson’s.
Understanding these four levels of organization is fundamental to biochemistry, pharmacology, and the future of synthetic biology.